This issue is brought to you by Quantum Leads. If you’re ready to stop chasing prospects and start closing high-intent deals, we’ll build the precision pipeline you need to scale. Click here to supercharge your sales today.
I’m thinking about publishing more video content on Substack.
I would love your feedback. Do you prefer my written newsletters? or do you also want to see more video content like this? I’m trying something new so I genuinely would love your feedback.
Hit reply and let me know.
Enjoy. :)
Section 1: What Are Stem Cells?
Why They Are Unique: Stem cells are the body’s raw materials. They are unique because they can divide repeatedly to renew themselves and can differentiate into functional cells to replace damaged or diseased tissue.
The Core Science: When a stem cell divides, each new cell has the potential either to remain a stem cell or become another type of cell with a more specialized function, such as a muscle cell, a red blood cell, or a brain cell.
Current Proven Uses: They are not just “theoretical” science; they are currently used in life-saving therapies like bone marrow transplants to treat blood cancers and other immune disorders. (Source: National Institutes of Health)
Section 2: The “Dark Ages” of Regulation
The FDA Stagnation: Historically, the FDA classified a patient’s own processed cells as “drugs,” requiring decades of clinical trials and billions in funding.
The Panama Pipeline: Due to US restrictions, a massive “Stem Cell Tourism” industry emerged. Patients often traveled to Panama, Mexico, and Colombia, spending $20,000–$50,000 for treatments for autoimmune and orthopedic issues that were unavailable domestically.
The Risk Factor: US doctors operated in a legal “gray area,” often risking their professional standing to provide autologous (using the patient’s own cells) treatments.
Section 3: The “Freedom Laws” (The 2025-2026 Tipping Point)
We are witnessing the “Cannabis-ization” of stem cells: states are bypassing federal bottlenecks to create “Medical Sanctuary Zones.”
Wyoming
SF 0048: Stem Cell Freedom Act
Signed March 2026
The most aggressive law in the US. It prohibits the Medical Board from disciplining doctors for using autologous (own) cells and prevents state officials from blocking patient access.
Utah
Signed March 2026
Provides a legal “Safe Harbor” for clinics. As long as specific disclosures are posted, doctors can perform therapies without fear of “unprofessional conduct” charges.
Florida
Effective July 2025
Explicitly allows licensed MDs/DOs to perform stem cell treatments for orthopedics, wound care, and pain management.
Connecticut
Effective Jan 2026
Mandates insurance considerations for regenerative biomarkers and protects “Right to Try” protocols.
Texas
Enacted
The foundational law that first allowed investigational stem cell use for patients with chronic illnesses.
Section 4: The Economic Impact
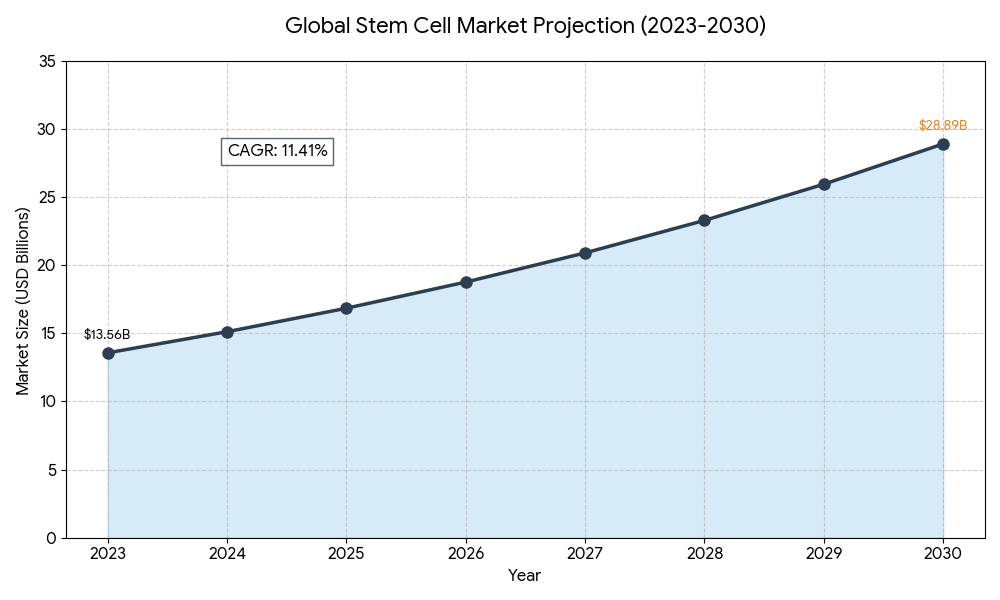
Market Forecast: The global market is projected to reach $28.89 Billion by 2030 with a steady Compound Annual Growth Rate (CAGR) of 1.41%.
(Source: Grand view research)
Global Stem Cell Market Projection (2023-2030)
The graph below illustrates the aggressive growth of the industry as state-level legalization removes barriers to entry.
Clinic Economics: A high-end regenerative clinic can generate between $150,000 and $400,000 per month in revenue. Because these are largely “cash-pay” procedures, margins often exceed $60%
The Buying Demographic:
The Aging Wealthy: 70+ million Baby Boomers looking for mobility and joint repair.
The High-Performance Athlete: Elite competitors using cells for career longevity and rapid injury recovery.
The Biohacker: 30-50 year-old professionals focused on “cellular optimization” and preventative aging.
Take hormone replacement
Take peptides
Do bloodwork
Section 5: 4 Business Models for the Stem Cell Era
1. The Growth Agency (Marketing)
The Model: Helping doctors bridge the gap between “Science” and “Sales.”
Example: A marketing agency that specializes in “Patient Education Funnels,” using webinars and long-form content to convert chronic pain sufferers into stem cell candidates.
2. Lead Generation / Concierge
The Model: Acting as a trusted “middleman” for patients seeking specialized care.
Example: A platform that vets clinics in “Freedom States” (like WY, UT, and FL) and charges a referral fee for every qualified patient they send for a consultation.
https://stemcellauthority.com/
3. The Lab & Distribution Model
The Model: Providing the “shovels” for the regenerative gold rush.
Example: Opening a GMP-Certified processing lab that partners with local surgical centers to harvest, process, and store (Bio-banking) a patient’s own stem cells for future use.
https://www.americordblood.com/
4. E-commerce / Ancillary Health
The Model: Selling consumer products that support the regenerative environment.
Example: A specialized supplement line formulated to “Prime the Niche.” Optimizing the body’s internal environment before a stem cell procedure—or topical post-care recovery creams. Very similar to peptides.
Important Disclaimer: This guide is for informational purposes only. Stem cell laws are evolving rapidly. Always consult with a healthcare attorney and a qualified medical professional before making investment or health decisions.